Join us:
See open positions

The story behind solid oxide technology
We believe combustion engines should be confined to the history books. When fuel is combusted, it emits harmful pollutants and greenhouse gases, whereas electrochemical conversion using fuel cell technology directly converts fuel into electricity and heat with low-to-zero emissions.
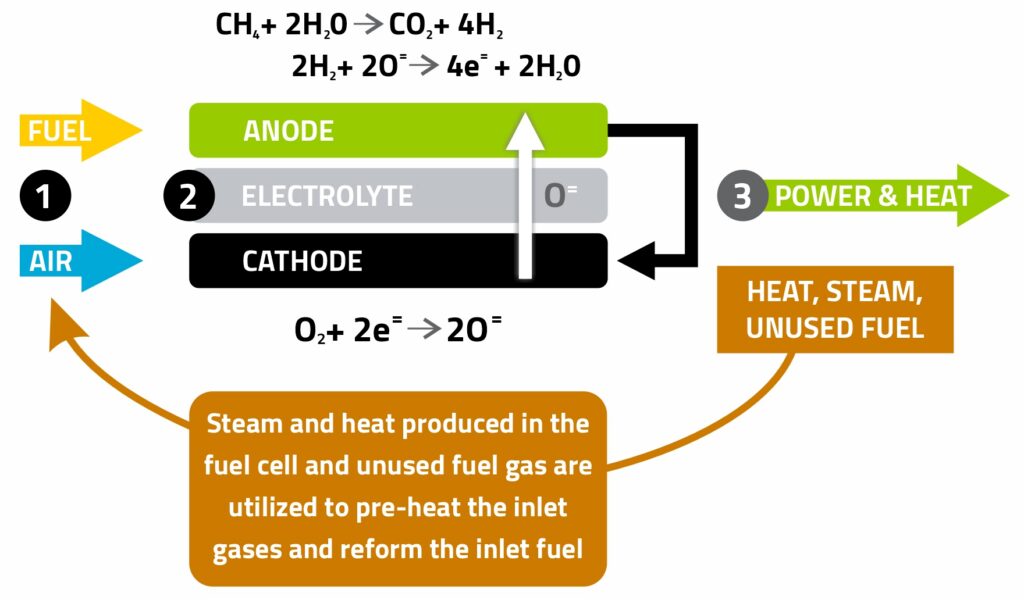
In a solid oxide fuel cell, air is supplied to the air electrode (cathode) and fuel to the fuel electrode (anode). When air comes in contact with the air electrode, oxygen ions are formed and transported through the dense, solid electrolyte. On the other side of the cell, the hydrogen-containing fuel gas sweeps over the surface of the fuel electrode and reacts with oxygen ions, producing electricity, heat, and steam.
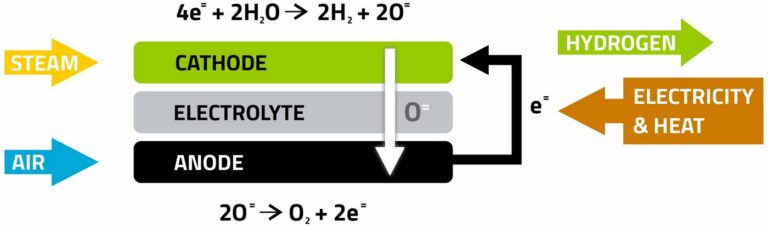
In solid oxide electrolysis, electricity derived from renewable sources is used to split steam into gaseous hydrogen and oxygen according to the reaction H₂O → H₂ + ½O₂
Electrolysis reaction occurs in an electrolysis cell, where hydrogen is produced at the cathode and oxygen at the anode.A solid ceramic electrolytes is used both to separate the gases and to facilitate the transport of ions necessary for the electrochemical reactions. Hydrogen production capacity can be increased by connecting multiple electrolysis cells in
series to form a stack.
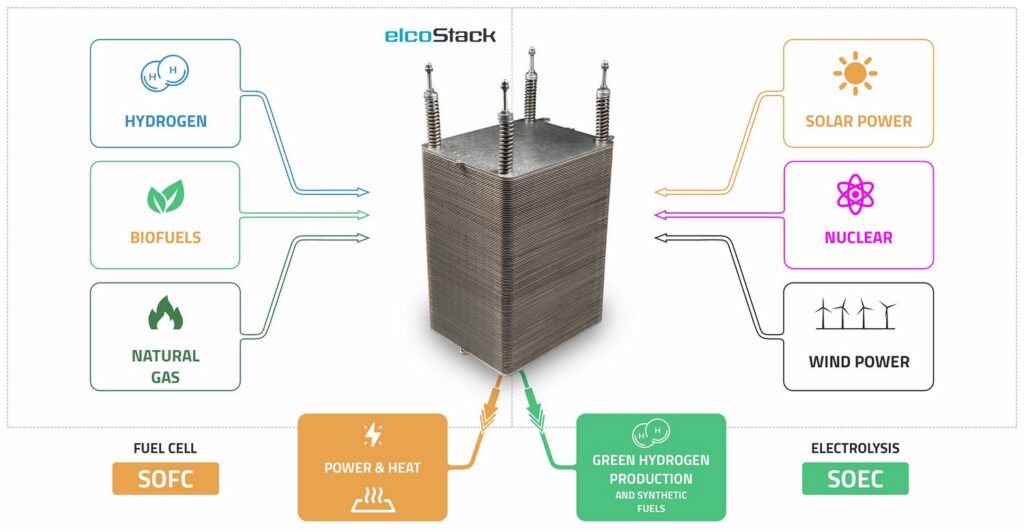
Its production methods, from fossil fuels to renewable energy, have very different impacts on carbon emissions: